4.2 Structure of the Nuclear Atom
Describe the structure of the nuclear atom. Section 42 Subatomic Particles Atoms are divisible into smaller particles.
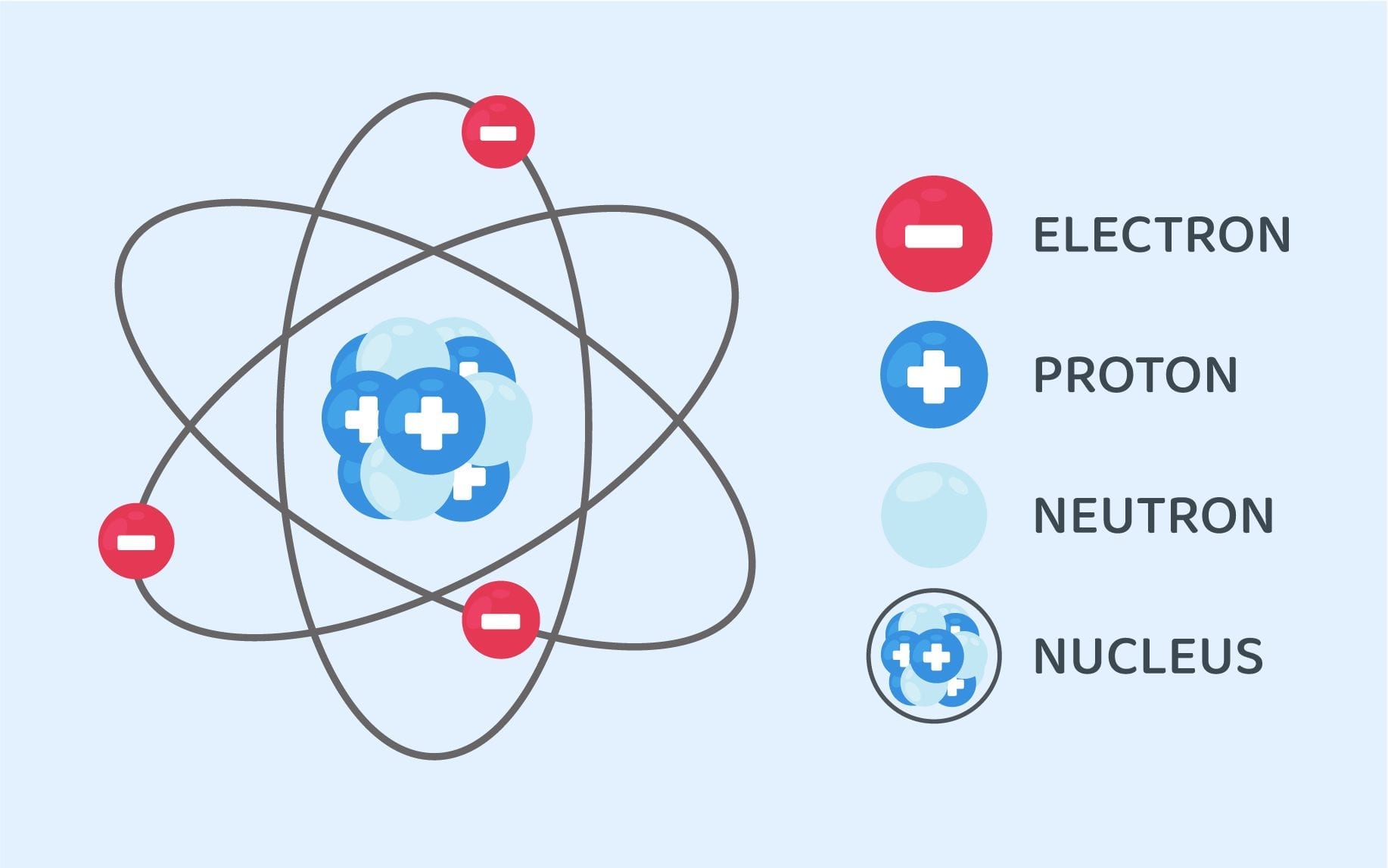
Structure Of An Atom Class 9 Science Notes Leverage Edu
Daltons atomic theory proposed that everything was composed of atoms that were invisible to the human eye.

. Read all tables and graphs. J J Thomson was the first one to propose a model for the structure of an atom. 9th - 12th grade.
Use the checklist below as a guide. Architect Last modified by. Chapter 42 Structure of the Nuclear Atom Subatomic Particles What are three kinds of subatomic particles.
With Super get unlimited access to this resource. At Grade Created by. For explaining this many scientists proposed various atomic models.
Understand the contributions of J. They can be broken down into even smaller more fundamental particles called subatomic particles. Read all section titles.
Distinguish between the three main subatomic particles. Thomson Robert Millikan and Ernest Rutherford to atomic theory. While all atoms of an element were identical different elements had atoms of differing size and mass.
Earnest Rutherford disproved the plum-pudding atomic model. Neutrons have no charge but mass of them almost equals mass of protons. Learn vocabulary terms and more with flashcards games and other study tools.
Chemistry 42 The Structure of the Nuclear Atom. Read all boldfaced words. Born in New Zealand Rutherford was awarded.
42 Structure of the Nuclear Atom 33 Copyright Pearson Education Inc or its affiliates. Structure of the Nuclear Atom Author. About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy Safety How YouTube works Test new features Press Copyright Contact us Creators.
42 42 Structure of the Nuclear Atom What evidence did Thompson have that led him to conclude the cathode ray was a stream of tiny negatively charged particles. The Nuclear Model of the Atom. The Structure of the Atom 47 Name Date cathode ray electron nucleus proton neutron concentrate The Structure of the Atom Section 42 Subatomic Particles and the Nuclear Atom Scan Section 2 of your text.
Three kinds of subatomic. The experiment that tested atomic structure was called the gold foil experiment. The Atomic Nucleus Rutherfords Gold-Foil.
The ray was attracted to a metal plate with positive electric charge and Thompson knew that opposite charges attract and like charges repel. Daltons atomic theory also showed that all compounds were composed of combinations of these atoms in ratios. 421 Thomsons Model Of An Atom.
Rutherfords atomic model is call the Nuclear Atom. It was then considered necessary to know how electrons and protons are arranged within an atom. Much of Daltons atomic theory is accepted today.
Knew that the mass of the atom was greater than its proton content. One important change however is that atoms are now known to be divisible. They inferred that this extra mass was due to neutral particles which were called neutrons.
MHHS Last modified by. Start studying SECTION 42 THE STRUCTURE OF AN ATOM. 9292006 42338 PM Document presentation format.
42 Structure of the Nuclear Atom Author. James Chadwick pro-vided evidence for neutrons in 1932. Section 42 Structure of the Nuclear Atom 105 Figure 45a shows how a cathode ray is deflected by a magnet.
Chemistry 12th Edition answers to Chapter 4 - Atomic Structure - 42 Structure of the Nuclear Atom - 42 Lesson Check - Page 109 12 including work step by step written by community members like you. Protons are positivly charged paticles. Electrons are negativly charged particles.
The nucleus is the tiny central core of an atom that is composed of protons and neutrons.
The Structure Of The Atom Boundless Chemistry

Atomic Structure 7 1 1 Edexcel Igcse Physics Revision Notes 2019 Save My Exams

Fundamental Particles 1 1 1 Aqa A Level Chemistry Revision Notes 2017 Save My Exams

Atomic Structure 11 1 1 Cie As Physics Revision Notes 2022 Save My Exams
No comments for "4.2 Structure of the Nuclear Atom"
Post a Comment